Anfahrt & Lageplan
Der Campus der Technischen Universität Dortmund liegt in der Nähe des Autobahnkreuzes Dortmund West, wo die Sauerlandlinie A45 den Ruhrschnellweg B1/A40 kreuzt. Die Abfahrt Dortmund-Dorstfeld auf der A40 führt zum Campus-Nord. An beiden Ausfahrten ist die Universität ausgeschildert.
Direkt auf dem Campus Nord befindet sich die S-Bahn-Station „Dortmund Universität“. Von dort fährt die S-Bahn-Linie S1 im 15- oder 30-Minuten-Takt zum Hauptbahnhof Dortmund und in der Gegenrichtung zum Hauptbahnhof Düsseldorf über Bochum, Essen und Duisburg. Außerdem ist die Universität mit den Buslinien 445, 447 und 462 zu erreichen. Eine Fahrplanauskunft findet sich auf der Homepage des Verkehrsverbundes Rhein-Ruhr, außerdem bieten die DSW21 einen interaktiven Liniennetzplan an.
Zu den Wahrzeichen der TU Dortmund gehört die H-Bahn. Linie 1 verkehrt im 10-Minuten-Takt zwischen Dortmund Eichlinghofen und dem Technologiezentrum über Campus Süd und Dortmund Universität S, Linie 2 pendelt im 5-Minuten-Takt zwischen Campus Nord und Campus Süd. Diese Strecke legt sie in zwei Minuten zurück.
Vom Flughafen Dortmund aus gelangt man mit dem AirportExpress innerhalb von gut 20 Minuten zum Dortmunder Hauptbahnhof und von dort mit der S-Bahn zur Universität. Ein größeres Angebot an internationalen Flugverbindungen bietet der etwa 60 Kilometer entfernte Flughafen Düsseldorf, der direkt mit der S-Bahn vom Bahnhof der Universität zu erreichen ist.
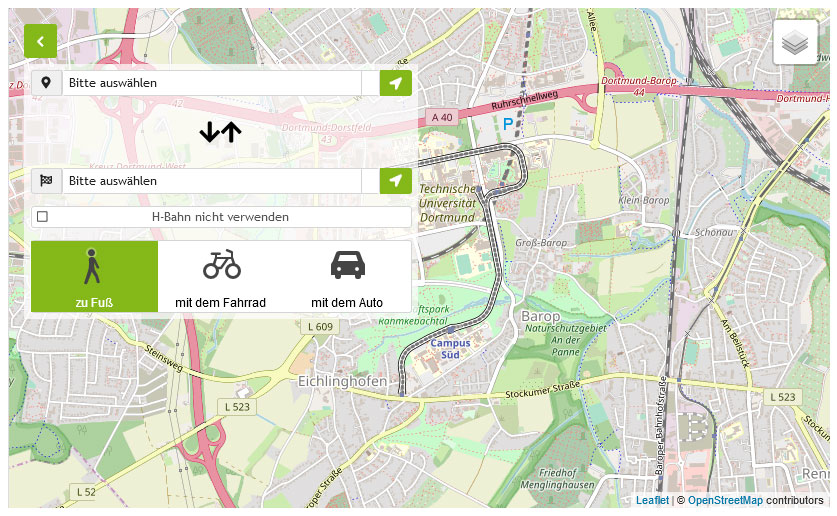
Lageplan & interaktive Karte
Die Einrichtungen der Technischen Universität Dortmund verteilen sich auf den größeren Campus Nord und den kleineren Campus Süd. Zudem befinden sich einige Bereiche der Hochschule im angrenzenden Technologiepark.
Meta-Navigation schließen